Copyright Alan Whitehead & Earthschooling: No Part of this book, post, URL, or book excerpt may be shared with anyone who has not paid for these materials.
Alan speaks in a very symbolic and esoteric manner in some parts of his books. Although they can be read anthroposophically, passages speaking of Atlantis, archangels, gods, etc. do not need to be taken literarily to be meaningful. The more you read, the more you will realize he uses many different religions to express ideas in a symbolic manner and not in a religious manner. His writings are not religious. In some places his writings are meant to refer to religious events in a historical way. In some places he is using religious figures (from Christianity, Judaism, Islam, Buddhism, Hinduism, Paganism, Ancient Roman and Greek Religions, etc.) in a symbolic manner. However, at no point is he promoting a specific religion or speaking from a religious point of view.
I have kept the writing as close to one-hundred percent original so you will also find that he speaks of Australia often and some spelling or manners of speaking may be cultural. Any words I have changed are presented like this: <word>.
Also keep in mind that these books are written by a Waldorf teacher with decades of experience who also studied with a Steiner student himself, so he speaks to an audience that is dedicating their lives to the Waldorf method without exception.
Because of this, all of his views are not reflected in the Earthschooling curriculum and not all of them may be ones you want to embrace or are able to use. In all of Alan Whitehead’s writings the opinions are his own and may not align with Earthschooling or Waldorf Books. In some cases, we will be updating some of these chapters in the future with additional and/or updated information.
Ultimately, however, as I read through these passages I find I can distill wisdom from even those paragraphs that do not resonate with me.
We invite you to read with an open mind and heart and with eagerness to learn and discuss…
THE DEED OF PROMETHEUS
Heat Physics Main Lesson – Class 11
In Steiner Education, the main lessons call on the head organization of the human being. The head was created – of the four cardinal organizations of head, chest, abdomen and limbs – on the first of Earth’s planetary incarnations, Ancient Saturn. As well as the human head, in its metaphysical or physics laws sense of course, the first of the four elements was created here, that of Heat. Old Saturn was indeed a heat globe; a big, fiery ‘head’ comprised of all today’s humanity’s smaller ones. Indeed the head is still the center of the human heat organization, as infra-red photography revels.
This main lesson-heat partnership suggests it as the optimum lesson in the 3-fold day (morning mains, meddles, afternoon blocks) in which to teach a 3-week Science unit on Thermodynamics or Heat Physics.
In the great evolutionary panorama described so vividly by Rudolf Steiner, we next meet the phenomenon of heat in Polaria. This was a recapitulation of Old Saturn, the first of the ‘epochs’ of our material planet earth (we are in the 5th epoch, the Aryan). Greek visionaries personified this period of now physical heat with the deeds of Prometheus, he who ‘stole’ the fire from heaven, from pre-Polarian, purely spiritual conditions.
Fire on Polaria was now not only physical, but retained elements of its spiritual origins in relating to the creative spark in the human soul. Prometheus was, after all, the creator of mankind. For this great deed he first had to separate the four elements from their seminal state of Chaos; the fiery element forming the sky. This productive principle reflects the stream into which Heat Physics is taught. This is Science – the fourth of the four main lesson streams – that relating to the Physical Body (Language-Ego, Maths-Sentient, Social Science-Life). The physical body was, as the Akashic Chronicle unveiled to Rudolf Steiner, also created on Ancient Saturn, and materialized – still as heat only – on Polaria.
Polarian Prometheus was a Titan; his gift of fire to humanity (no animals use fire) was an incandescent torch lit from the Sun Chariot. As a Creator, he was also the Good of Science. A closely allied principle is found in the Hebrew Cabbalistic interpretation of the same phenomena, this time as Adam. Man’s ‘father’ is the Cancer representative (Adam means ‘red earth’ – the color of both Old Saturn and Polaria was nominally red) in the Hebrew Zodiac. The Crab is the sign informing and inspiring Science in the esoteric mystery that is Steiner Education’s Subject Zodiac.
So, fire, or heat, is all to do with first principles, of Creation. It is good to begin the unit with a myth or three (there are many) to place this conceptually-demanding subject into an imaginative framework. Historically, fire was mastered as a tool of civilization in the great Persian (Khmer in the East) era. This was the 2nd post-Atlantean Civilization of the Aryan epoch (our own Anglo-Nihon is the 5th). In Persia-Khmer, Heat Physics was applied in a practical way in cooking, pottery and even smelting – of soft metals at least, like gold, silver, tin, lead and copper.
This was elaborated upon in the Bronze Age of Egypt (Han China); and further still into the Mars-ruled Iron Age of Greece and Rome (Khan China). One massive pure iron column from India has baffled scientists trying to explain its total resistance to rust; a feat which cannot be replicated with the most sophisticated technology today.
Heat Physics, in a formalized sense at least, had to wait until the dabblings of the Medieval alchemists for a new impulse. This led, a century or so later, to the explosion of knowledge in the scientific age; all based on thermodynamics, First Principle of physics.
There was a marriage of theory and practice in the 19th Century, when man harnessed the power of fire to drive machinery. At first this was relatively simple coal-fired steam engines; then there was oil – and today, nuclear. Tomorrow – hopefully – the power of sun heat will generate the energy modern man thinks he needs for very survival.
The mission of science in the new millennium is to return the torch of the Sun Chariot via the spiritual implications inherent in solar energy.
In the great recapitulation of the developing human being, 17-year-olds are experiencing the 19th Century period (beginning Ancient Saturn in the first year of life). As such, there is no better time to teach Thermodynamics than in this Industrial Age year. Without the 19th Century mastery of fire on a grand scale, the immense technological advances of the 20th Century would not have been possible. Hence Class 11 students are perfectly positioned to revisit this amazing scientific 19th Century awakening.
Fire or heat is of course present – sometimes active by its absence – in all phenomena; but to simplify the created world so that it can broadly be comprehended, we find Seven Realm which are bestowed life by fire. The first is Pyrosphere; a high-altitude envelope of intense heat surrounding the earth. The task of the Pyrosphere is to burn up any cosmic invaders, like space rocks. Re-entry space craft, like the famous Shuttle, are equipped with a special ceramic nose cone to survive tis perilous passage. The Atmosphere is dominated by heat, which creates the dynamics of weather – ditto for the Hydrosphere, its task is keeping the oceans moving, mainly through changes in temperature.
Without the continual displacement of warm and cold water, in both horizontal and vertical currents, there would be a deathly stillness in the world’s deeps and shallows. The Petrosphere is a very self-evident fire zone, especially in volcanic areas. Fire truly is the crucible from which the crystalline earth is created. The Plant Kingdom too is a generator, conserver and absorber of heat. An Arctic forest is warmer than its equivalent bitter, windswept tundra. Animals are not only all the above, but actually creators of heat; especially those with warm blood.
Finally the 7th heat Realm, from the high heavens all the way down to Man. Unlike all other six realms, we are proactive in heat creation, from igniting a simple campfire (which no animal can do) to creating a nuclear’ bomb.
In short, we have become as gods, sadly with the ability to destroy far greater than that to create. We are like Agni (akin ‘igni’, as in ignite), the Hindu fire deity – or the Salamandas, Meliades, Min Min or Hobs, ubiquitous fire spirits the world over; or, to return thence we came, to Pyrrha, daughter of Pandora, sister-in-law of Prometheus.
The following are just a few words to which Pyrrha (from ‘pur’, fire) gave her name: pyrrhic rhythm – a Greek war dance; pyretic – fever; Pyrheliometer – instrument for measuring sun heat; pyrite – mineral for striking fire; pyrolysis – arson; pyrophobia – fear of fire…and so on!
These kinds of classical or mythical allusions are used right through the 3-week unit in order to ensoul the otherwise lifeless content. Though the lesson has a head start in being programmed in the Physics (where else?) strand; the first of three in the Science stream (the other two being Chemistry and Biology). This triune calls on the three soul forces of will, feeling and thinking (or on a higher plane, body, soul and spirit).
Will, in esoteric understanding, is related to fire – “The fire of the will.” As the saying has it. The will, by its nature, demands a certain hands-on expression; a good starting point might be small demonstrations or even experiments with the various Temperature Indicators. These include special tapes which turn lighter and darker according to the temperature; paint that does likewise; forge workers determine the approximate temperature of a metal by its color, right up to ‘white heat’. Then there are ceramic cones and melting pellets used by potters and ceramicists. Next are crystals, which are very accurate, some to 0.1°c.
From indicators, we lead naturally on to heat measurement proper; a study of the more common thermometers. Where possible, have examples in the classroom. Most of these are based on the physics of Kinetic Energy, where heat application excites molecular movement. This it is which expands the mercury (or alcohol) in the common thermometer, sending it up the glass tube.
The Mercury Thermometer is only suitable for measuring temperatures from -30°C to 500°C, due to quicksilver (the movement metal par excellence) freezing at -39C and boiling at 357°C (both at one ‘atmosphere’ – an ATM). These top and bottom limits, known as fixed points, herald a Change of State in an element; as in a liquid turning to steam at the top, and becoming a solid at the bottom.
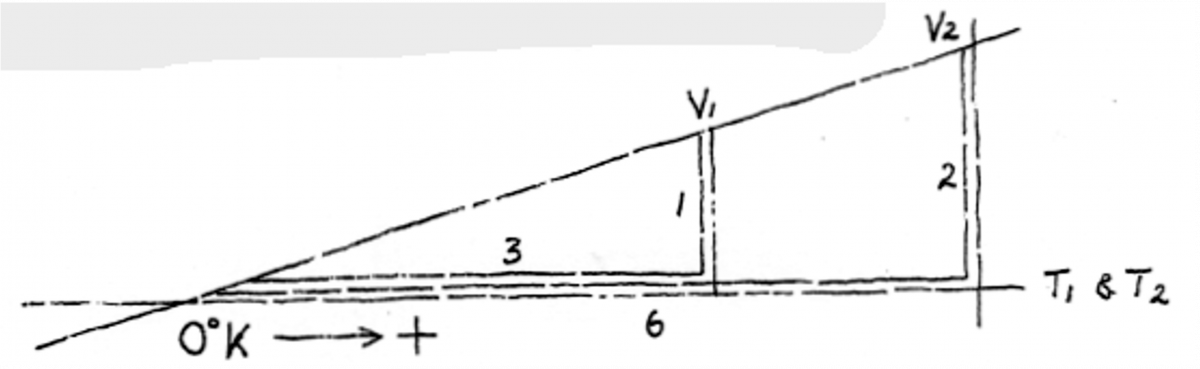
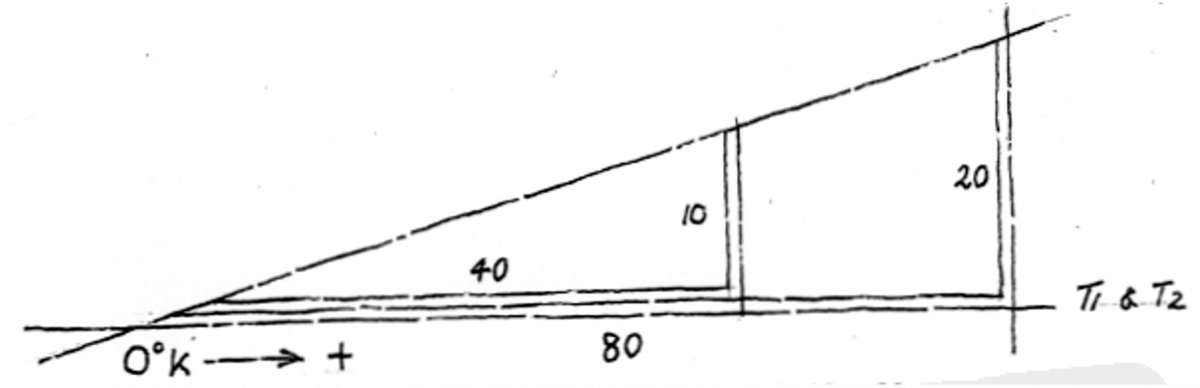
Exercises in changing Fahrenheit to Celsius are useful. In all scientific areas other than meteorology, Celsius is preferred to centigrade due to the latter’s possible confusion with a hundredth of a grade (as in angle measuring). Celsius is named after the 18th Century Swedish astronomer Anders Celsius. Due to it being a proper noun, like Fahrenheit, it is always capitalized. So, Fahrenheit to Celsius: merely subtract 32 and multiply by 5-over9 – e.g. 215°F-32 = 183-over-1 times 5-over-9 = 101.6C. Now C to F: add 32 then divide by 5-over9: e.g. 183°C+32 = 215-over-1 divided by 5-over-9 = 387°F.
There are always three heat measuring systems, the third being Kelvin. Fahrenheit however is largely phased out in Australia, Celsius today being every person’s measuring scale – while Kelvin is exclusively used by the scientific community. William Kelvin was the 19th Century British thermodynamicist who introduced the system.
Kelvin is one of the seven official System International (SI), the other six being meter, kilogram, second, ampere, candela and mole (the seven planetary aspects of material creation perhaps?). Not that you need to know this, but a Kelvin is 1-over-273.6 of the thermodynamic temperature of the triple point of water; symbol capital K.
Mere mortals, non-scientists, would never be required to measure the lowest temperature known, or conceived of at least (actually no-one has, this is the Holy Grail of thermodynamics!), which is
-273°C, or 0°K, or 0°A; the last representing Absolute Zero. This is the state of cold in which it is thought that all molecules of any substance stop moving altogether. Another enigmatic speculation is that if this state is actually obtained, the substance would vanish – would become anti-matter!
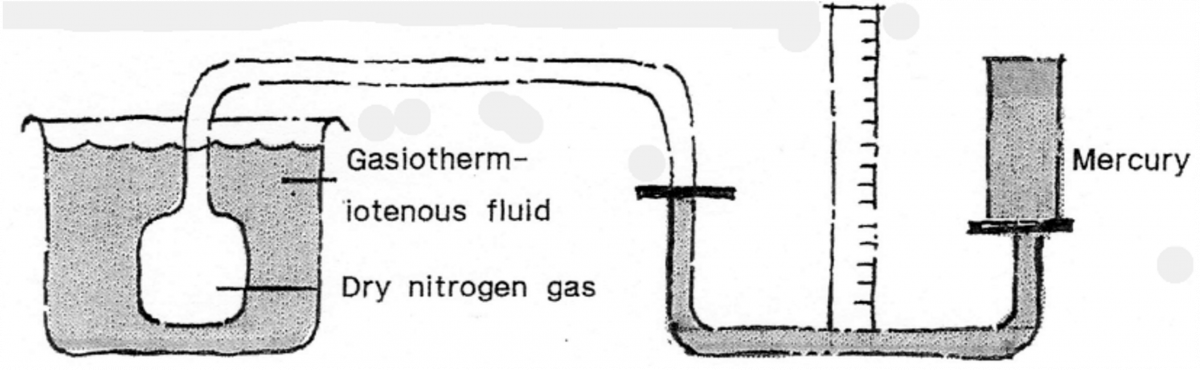
The word ‘therm’ is from the Greek meaning heat. One therm is a British unit of heat equal to 100,000 thermal units, or 1.055056×10 to the power of 8 joules (a joule is a unity of energy). Bat back to thermometers; there is also a Gas Thermometer, usually suing a dry gas, like nitrogen. This is combined with mercury and is used to accurately measure the temperature of liquids where the measuring end is separated from the scale, as in a sealed tank.
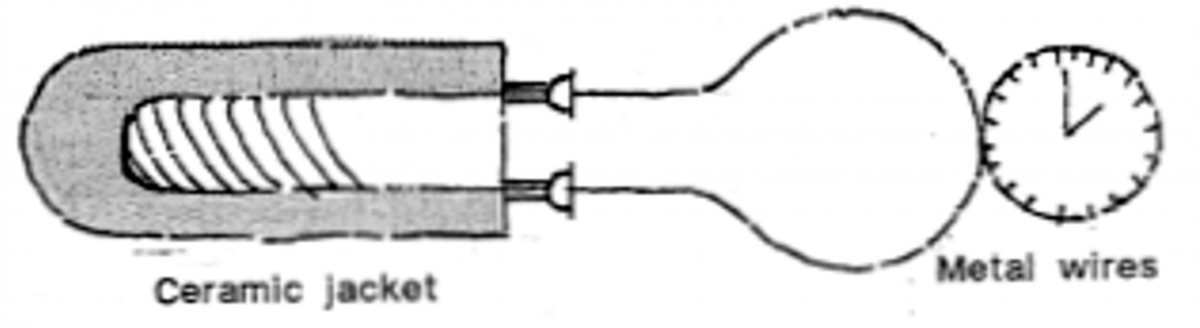
A Resistance Thermometer is based on the physics of the temperature of a metal (in this case wires) rising as its electrical resistance does. Again the thermometer can be some distance from its subject – and can measure very high temperatures, as found in a furnace. This can be accurate to 0.0001°C. A variation, the Bolometer, can even measure star temperatures from telescopes.
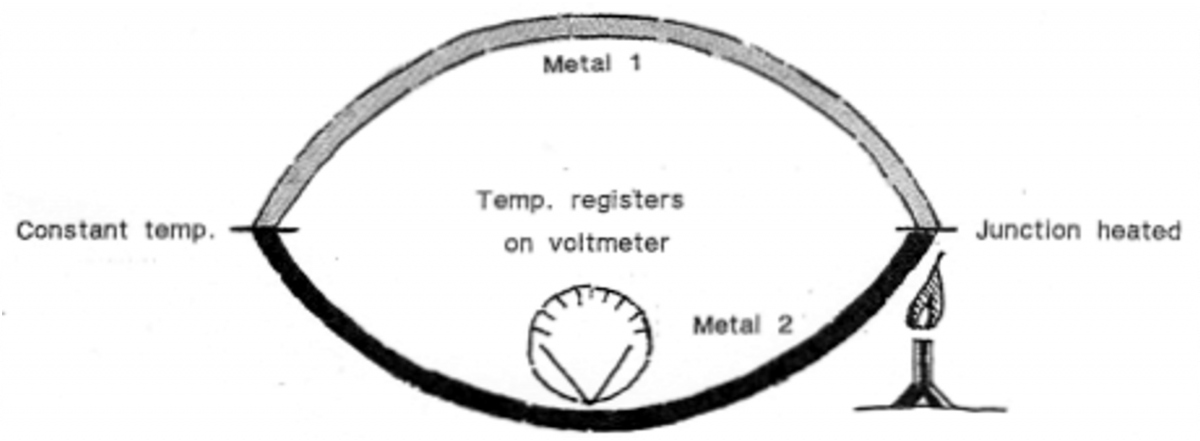
The Thermocouple also uses metal but, as the name implies, two different kinds. These are usually iron and copper working at odds; as Mars and Venus so often do. In this case the variation of the thermoelectric effect measures the temperature.
This has the widest measuring capacity of all, from 1°K to 3000°C – from fine readings like skin temperature, to the interior of a steelworks coke oven! The difference in temperature of the two metals creates a flow of electrons (net e.m.f.) which registers on a voltmeter.
The Radiation Pyrometer is only for measuring objects over 600°C. It is based on the fact that infra-red radiation is emitted, which travels through space; hence no physical contact with its subject is necessary. These heat rays neither express through conduction nor convection. There is also a continuous recording capacity; useful in measuring the heart of a volcano for instance, as they routinely do.
But back to the mundane, with the common thermostat, or Bimetal Thermometer as it is officially called. This is found in refrigerators and other household and industrial appliances. Try and have one in the classroom to demonstrate its simple but clever physics. Again these use two opposing metals, and are very rugged, the kitchen fridge being one of the most reliable and long-lived of appliances. The two metals are often Invar (iron-nickel alloy) and brass. When the metal expands, due to increasing heat, the system activates; as the temperature drops, as detected by an air-conditioner, the second metal contracts to shut it off again.
The Acoustic Thermometer returns us to the arcane; this is based on the law that the velocity of soundtraveling through a medium is dependent on its temperature.
The Magnetic Thermometer is used for the lowest possible readings, down to 0.002°C! This is based on the principle that the magnetism of a material is determined by its temperature.
Finally, but not comprehensively, we have the Thermal Noise Thermometer: static sound varies with temperature change due to increased motion of electrons.
Thermography is the science of mapping topography with infra-red film; this photographs not the structure of the land surface, but its temperature. Infra-red photography is also used in biology, medicine and legion other scientific disciplines. It provides not only different information than optical photography, but more; due to infra-red being a far broader spectrum than light.
On the opposite end of the light spectrum is ultra-violet, the cold band. Various phosphers found in many minerals, plants (phosphorescent fungi) and animals (fireflies and some corals), when exposed to UV light emit variable light intensities, dependent on their temperature – being brighter as the mercury rises.
Thermography is the science of mapping topography with infra-red film; this photographs not the structure of the land surface, but its temperature. Infra-red photography is also used in biology, medicine and legion other scientific disciplines. It provides not only different information than optical photography, but more; due to infra-red being a far broader spectrum than light.
On the opposite end of the light spectrum is ultra-violet, the cold band. Various phosphers found in many minerals, plants (phosphorescent fungi) and animals (fireflies and some corals), when exposed to UV light emit variable light intensities, dependent on their temperature – being brighter as the mercury rises.
This is why so many phosphers, especially flora and fauna, are tropical. The thermodynamic polarity of infra-red and ultra-violet express the power of Aquarius and Libra respectively; for more information on this color-cosmic phenomenon, see my book A Spiritual Science.
If Thermometry is the science of heat measurement, then Pyrometry is that of recording same, especially very high heat, especially radiation. Radiation Thermometry is heat radiated from a substance or object. Galileo built, in 1592, the prototype of today’s sophisticated heat measuring instruments, his simple but effective Thermoscope. This measure not temperature itself, but the change in temperature. After all, temperature is not a quantity, but a quality; the temperature of the tea in the cup and in a teapot may be the same, but there is much more heat – a quantitative property – in the latter
One of my students was more interested in drawing the tea set
than in the thermodynamics it represents!
Heat, although one of the classic 4 Elements, has no molecular structure. On this basis some scientists disclaim its existence as a separate entity at all, regarding it merely as a condition of substance. After all, heat cannot be measured or perceived in any way until bonded with matter of some kind; whether gas, liquid or solid. This is dependent on the object’s molecular structure (‘installation capacity’), surface texture and color. Other experts assert that heat – as a separate entity – does exist, but as plasma (meaning to mold – akin plastic). Plasma is ionized material consisting of a nuclei and electrons; and thought to be a major component of the sun, most stars, and fusion reactors.
They refer to plasma, or primeval energy, as the 4th State of Matter. Spiritual Science has it as the First! In fact in Steiner’s view; warmth and warmth either (‘to glow’) are two expressions, one physical one metaphysical, of the same phenomenon – the edges were one becomes the other being frustratingly blurred to his spiritual researcher!
“Plasma is like a fourth state of matter. In a vacuum environment where you have strictly gases and high energy for example, a lot of light coming out of the sun splits the atoms into free electrons, ions, neutral atoms and other forms of energy, like stored magnetism, stored electrical fields – that’s plasma.” James McCanney, Electro physicist, Cornell University.
Temperature is also the only sibling of the 4 Element offspring which in ‘intensive’; the other three being extensive. For example in the latter, 1 meter plus 1 meter equals 2 meters. An intensive property is rather: 1 cup of 20° plus 1 cup of 20° equals 2 cups of 20°!
For global consistency there is the International Practical Temperature Scale, the IPTS (est. 1968). This was determined by taking account of the Four Laws of Thermodynamics:
- Conservation of energy; this assumes a given amount of heat energy in the universe. The capacity of matter to conserve or release this is constant and measurable.
- Heat flows from higher to lower temperatures until equalized between the two. This is ‘thermal equilibrium’.
- Absolute Zero (as mentioned earlier) – but oddly there is no heat maximum, not even theoretical?!
- Law of Zeroth; no, not an exotic comic book character, but if temp. A = B and B = C, then A = C. Self-evident, yes?
While on more cryptic temperature measurement, there is also the British Thermal Unit, the BTU. This is the amount of heat required to raise one pound of water by 1°F at 1 ATM. 1 BTU is 256 calories. This is not only the frequency of middle C on the music scale, but 4 to the power of 4 – the ultimate earth of Physical Body number! It is good for young adults to be exposed to these more esoteric concepts.
The Linear Expansion Co-efficient of perfect gases are all the same, i.e.; they expand at the same amount with the same degree of heat. The LEC is the measurable increase of a substance, the amount it will expand under heat application – that is, its unit length increase of 1°C rise. For example iron has a LEC of 11 – 11 over 1,000,000 or 0.0000011, or 11 X 10 to the power of minus 6 = 11 – Whew! This means that a rod of iron under a 1°C increase = initial length X LEC X temp. rise. i = L X LEC X tr.
Here are a few examples for classroom exercises:
How much does a rod of iron 2.5cm long expand with 300°C temp. increase?
i = 2.5 X 11 X 300 = 0.00825M = 8.25mm
How long extra will a rail line of 15m be with an increase of 30°?
i = 15 X 11 X 30 = 4.95mm
What tolerance has to be built into a cylinder to accommodate a brass piston (454) of 80mm which increases temp. by 120°? (i.e.c 19)
i = 80 X 0.000019 X 120 = 0.1824mm
How much will a 1mm silica crystal (i,e.c. 0.5) lengthen with a temperature increase of 20°?
I = 1 X 0.0000005 X 20 = 0.00001mm
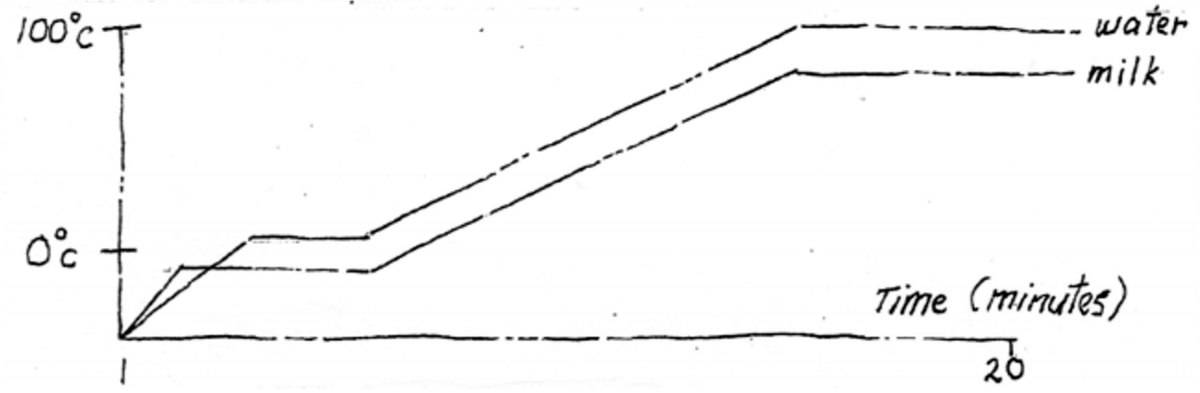
This well-ordered principle is in ironic, as it were, contradiction to the word gas, which means ‘chaos’. When a gas or vapor transmutes to pure energy, or warmth, it is said to contain ‘latent’ rather than ‘sensible’ heat. Liquid to vapor is Heat of Vaporization’ solid to vapor is Heat of Sublimation; and solid to liquid is Heat of Fusion. These mysterious transubstantiations are called changes of state.
As mentioned, heat energy absorption is dependent on molecular structure, e.g. iron absorbs heat 10 times faster than water. Hence a 1 kilo iron saucepan will heat up quicker than its 1k water contents. The iron will be 200°C while the water is still only 20°! Heat energy also changes molecular structure, again using water as an example. At 0° water becomes ice, at 1 ATM it boils at 100°, but on the top of Mt. Everest, at 0.75 ATM, it boils at a cool 75°!
As it can be seen, pressure affects heat energy (thus change of state); high pressure prevents molecular escape, low pressure (suction) assists it, in this case allowing the water to boil more quickly. Heat energy also creates changes in properties of substances; 1. Less density. 2. More viscosity. 3. Increases chemical reactions, and times. 4. Volume increases. 5. Increases chemical decomposition. 6. Increases electrical conductivity.
A Calorie is a volume of measurement for heat determined by the amount of heat energy required to raise 1gm of H20 by 1°C. The thermal capacity of a substance is the number of calories required to raise the temperature of 1gm of water per degree C. Iron is 1 calorie per gram per 9°C, and mercury is 30°. The thermal capacity (specific heat of substances) of some elements are water 1, air 0.24, glass 0.16, iron 0.15, copper 0.09, mercury 0.033.
The formula for finding the quantity of heat required to change the temperature of an object of known mass and TC through a known temp. change is heat (H) = mass (M) X TC X temp. change (T). How much heat must be applied to increase the temp. of 2kg of iron by 20°C? H = 2 X 0.105 X 20° = 4200 calories. Give exercises on the other substances mentioned, changing the values.
To find the TC of a substance of known mass and temp., place in water of known mass and temp. – find TE (thermal equilibrium) then apply formula: (substance) M X TC C T1 = (H20) M2 X TC2 X T2. For example 750gm of substance at 100°C placed in 200kg water at 25°C; therefore TE = 44°C. 750 X TC C 56 = 200 X 1 X 19 give TC X 42,000 = 3800 therefore TC = 3800 over 42,000 = 0.09 (copper).
Here are some basic temperatures for comparison (all in Celsius): that obtainable in a laboratory
-273.002°; polar weather -88°; comfortable for us 22.8°; torrid weather 58°; gas flame 800°; oxy-acetylene 2,500°; arc welding 6000°; nuclear reaction 20,000,000°!
Changes of Heat The latent heat of vaporization of a substance is the quantity of heat needed to change 1gm from liquid to vapor without change in temp.
LHV escapes with gas molecules. Water, one of the highest, takes 540 Cals. Per gm. It takes 100 Cals. To heat 1gm of H20 from 0° to 100°, and another 540 Cals. To convert it to steam – 6 times more. Steam burns more than water (all things being equal) as it contains the LHV which is released when gas condenses. This fact is exploited in steam room heaters, as steam transports more easily.The latent heat of fusion of a substance is the quantity of heat needed to change 1gam from solid to liquid without change in temperature. The LHF of ice is 80 calories per gram.
3 Laws of Temperature and Gas: Law 1 (Boyle’s) – at constant temperature, the volume of a gas varies inversely as the pressure upon it.
E.G. P1 V1 = P2 VP = 2 X 2 = 4 X 1 = 4
If a Scuba lift bag had 15 liters of air at 2 atmospheres, how many liters would it have at the surface?
15 X 2 = 30 X 1 Vi = 30 liters.
Law 2 (Charles’) – at constant pressure, the volume of a gas varies directly as the temperature V1 over T1 = V2 over T1
Law 3. At constant volume, the pressure of a gas varies directly as the temperature. P1 over T1 = P2 over T2
E.G. P1 over T1 = P2 over T2 = 10 over 40 = 20 over 80 – .25
SEVEN HEAT ENERGY SOURCES
According to the 1st law of thermodynamics, heat is preserved and can be released by an activator, there are sever of these:
- All biological metabolism releases heat energy, often through changes of state. Solids become liquids and vice versa.
- All combustion (burning) creates heat – this is caused by released gases reaching ignition temperature. No solid or liquid can burn. Non-organic substances tend to melt, organic ones to char; one turns liquid, the other solid.
- Electrical energy creates heat by the activation of molecules creating friction. This occurs when the electricity current is opposed.
- Mechanical energy excites surface molecules by friction.
- Oxidation creates heat, e.g. iron nails rusting in an enclosed container will raise the temp. inside.
- Chemical reactions create heat, some violently, like sodium bicarbonate and hydrochloric acid.
- Nuclear reaction occurs in fusion, as in a change of hydrogen to helium, or fission, changing uranium to lighter atoms, splitting the atom.
Liquids expand regularly with temperature rises, but at a different rate for different liquids.
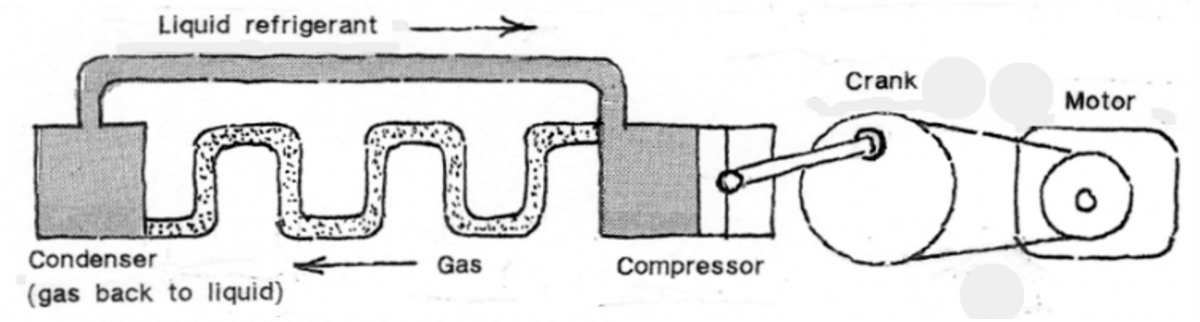
Diet calories are really Kilocalories, which are determined by the amount of heat required to raise the temperature of 1kg of H20 by 1°C (14.5° – 15.5°). Heat loss occurs through evaporation; local anesthetic works by applying ethyl chloride (a low boiling chemical) which immediately freezes on contact with the skin. Refrigeration works by this method.
Because of the foregoing principle, air temperature is lower around evaporating bodies of water, like pools, and the sea even.
Heat occurs when a gas condenses; the air is actually warmer when humid air condenses into rain or snow. Similarly, melting snow or rapid evaporation causes a drop in temperature. The co-efficient of expansion of perfect gases are almost the same; i.e. they expand at the same amount with the same degree of heat.
SOME HEAT TERMINOLOGY
Thermesthesia is sensitivity to heat. The rattlesnake hunts with the aid of two heat receptor organs in its nose, and only with these (at night anyway). The human’s most thermesthetic organ is the tongue. Some dying if there is only a 1° temp. change in their environment. They are usually more prolific in the tropics, but with the phylum’s status as having the greatest Thermoanesthesia as well, tolerance to cold, they are found in polar waters as well.
The humble mollusks are, after all, the animal with the first – albeit cold – vascular system; the first true blood. This can not only conserve heat better than the chemical equivalent that went before, but be more temperature sensitive. In pre-mollusk animals, heat is detected with Chemoreceptors; with mollusks and above, with Thermoreceptors. These are spots on the skin which are biophysically specific – the nerves onlyperceive heat or cold. The cold sports are continuously active electrically.
The central system for heat perception in warm-blooded animals (birds and above) is the head. This can be seen in mound builders, who poke their head into their leafy nest pile to gauge, to a very fine degree, the internal heat. However the mollusks are the animal phylum nominated by Spiritual Science as bearing the Sense of Warmth. A limpet can tolerate the greatest temp. range of any animal. Whales have a great range as well, while sharks must migrate on currents which retain stable temperatures.
Thermokinetics is the ability to heat to move; this is determined by the nature of the substance the heat in moving through, and the quality of the heat. Three main aspects affecting the latter are conduction – a hen sitting on eggs, convection – water circulation in a kettle, and radiation – the heat from the sun. These also affect Thermotropism, which is movement created by heat, like steam rising.
All living beings have a minimum and maximum temperature resistance, and a different optimum tolerance. With bees their in-hive optimum is 35°C, while their range is a tiny 1°C. Cold-blooded animals cannot achieve Homeostasis, a stable body temperature irrespective of outside temperature. They are Poikilothermic, while their warm-blooded peers are Homoiothermic!
Optimum growing temperatures for plants are often curious, the following is for garden peas.
Sweet pea flowers die before spring at temperatures above 20°C. Cherry trees will not set fruit without a lengthy exposure to winder cold. Potato tubers are controlled by night temperatures – optimum 10°-14°C.
Animals have a wisdom-filled arsenal for combating debilitating ambient temperature: burrowing; nests; hollows; incubation; hibernation and its summer equivalent aestivation; snuggling; dens; water; mud; shade; nocturnalism and diunalism; migration. In relation to keeping warm or cool in water, a Thermocline is often exploited.
Autumn tadpoles do not become frogs until Spring, yet Spring taddys become frogs after a very short gestation. Temperature extreme responses of animals take many forms: panting; open or closed pores; molting; sweating; circulation with wings; open moth; like writs (kangaroos); move; shiver; curl up; sprawl; wave ears; thick fur or nakedness. The Deed of Prometheus, ranging across all the natural sciences as this short expose describes, is more extensive than would at first appear!
Modern heroes, like the Volunteer Bushfire Brigade, don’t have to carry guns!

















Leave a Reply